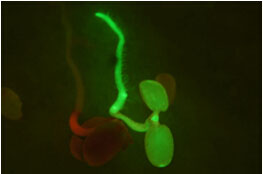
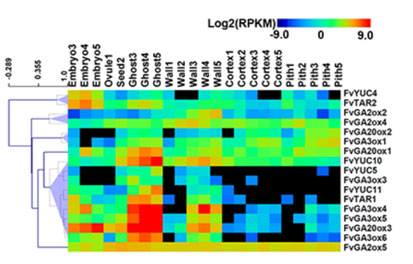
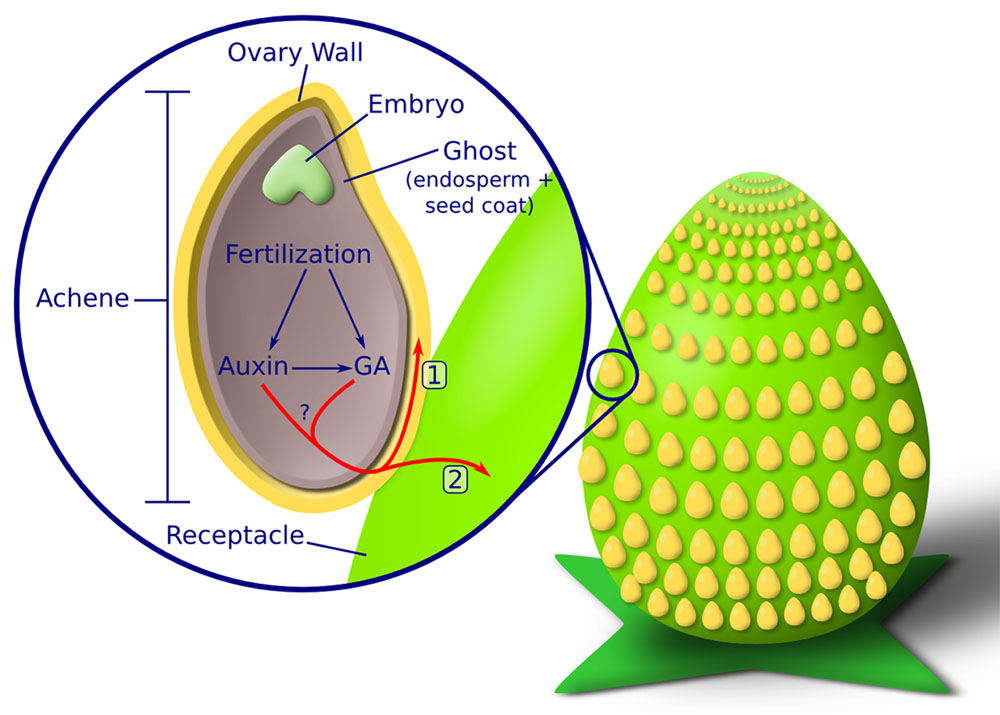
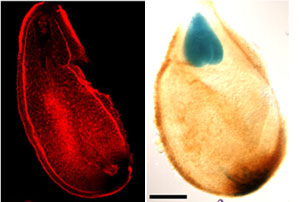
Flower and fruit are major contributors to food and feed. Our lab is interested in understanding the basic mechanisms controlling flower and fruit development as well as translating that knowledge into solutions for agriculture. We use two different plant systems, wild strawberry (Fragaria vesca) and Arabidopsis thaliana.